For research use only. This reference describes tirzepatide strictly as a chemical compound used in cell-based receptor pharmacology and animal research. Research-grade tirzepatide is not equivalent to, and is not a substitute for, the FDA-approved pharmaceutical product of the same chemical name. Nothing here is intended for human or animal consumption. We make no health, performance, or therapeutic claims of any kind. For full context, see our research-use disclaimer.
Tirzepatide — development code LY3298176, marketed by its sponsor under the brand names Mounjaro and Zepbound — is a synthetic 39-amino-acid linear peptide built on a glucose-dependent insulinotropic polypeptide (GIP) backbone. It engages two Class B G-protein-coupled receptors as an agonist: the GIP receptor (GIPR) and the glucagon-like peptide-1 receptor (GLP-1R). What follows covers the peptide's primary structure, the two non-coded residues that give it protease resistance and tuned receptor activity, the cryo-EM evidence for how a single continuous α-helix engages both receptors in distinct conformations, the imbalanced and biased pharmacology at GLP-1R, the structural class it belongs to, and the regulatory framing for laboratories ordering research-grade material.
Primary structure: a 39-residue GIP-derived peptide

Tirzepatide's chemistry is well-characterized in both the structural literature and standard reference sources. The compound is documented in encyclopedic references as a synthetic 39-amino-acid sequence built on a GIP backbone. The 2022 Nature Communications cryo-EM paper by Sun and colleagues is the primary structural reference for what follows. The structural-determinants paper by Coskun and colleagues isolates which positions in the sequence drive engagement at each of the two target receptors.
Why a GIP backbone
The choice of starting backbone matters for the same reason it does in other multi-receptor agonist programs. Among the targets, GIPR engagement is the harder problem when starting from native GLP-1; the GIP-derived sequence retains GIPR activity by default and leaves the medicinal-chemistry team free to tune the GLP-1R end. Earlier-generation GLP-1 analogs such as semaglutide and liraglutide already established the value of α-aminoisobutyric acid (Aib) at the protease-cleavage site. Tirzepatide carries that move forward into a dual-receptor frame.
Lipidation at Lys20
The peptide is acylated at lysine 20 with a C20 fatty diacid attached via a γGlu-2×OEG linker — that is, a γ-linked L-glutamic acid spacer followed by two 8-amino-3,6-dioxaoctanoic acid (OEG) units, terminating in the C20 fatty diacid. The fatty diacid enables high-affinity, reversible binding to serum albumin, which is the structural mechanism behind the long pharmacokinetic half-life seen in the literature. The same general lipidation motif appears across long-acting peptide agonists in the same structural class.
Engineered modifications: two non-coded residues
Two positions in tirzepatide carry non-canonical amino acids: α-aminoisobutyric acid (Aib) at positions 2 and 13. Each one earns its place for a specific structural reason.
Aib2 — protease resistance
Native GLP-1 and GIP get cleaved at their N-termini by dipeptidyl peptidase 4 (DPP-4). The enzyme cuts between residues 2 and 3 in both peptides. Aib at position 2 introduces backbone constraints that the DPP-4 active site can't accommodate, which is why the same modification appears in earlier-generation GLP-1 analogs. The structural-determinants analysis confirms that Aib2 is the principal source of protease resistance in tirzepatide.
Aib13 — GIP receptor activity
Aib at position 13 supports activity at the GIP receptor. Where retatrutide uses α-methyl-L-leucine at this position to support multi-receptor balance, tirzepatide uses Aib — a smaller, more conformationally-restrained substitution that mimics native GIP engagement at GIPR rather than introducing a wholly new pharmacology. The structural choice is "keep what works at GIPR, tune the rest for GLP-1R."
Lipidation chemistry as a structural-class motif
The γGlu-2×OEG-C20 motif on Lys20 is shared with several other long-acting peptide agonists in the same structural class. The γ-glutamyl spacer projects the linker out from the peptide surface; the two ethylene-glycol units add flexibility; the C20 fatty diacid provides albumin affinity. Researchers comparing tirzepatide to retatrutide or to semaglutide will see different specific positions and different fatty-acid chain lengths, but the same overall design language.
Receptor binding architecture
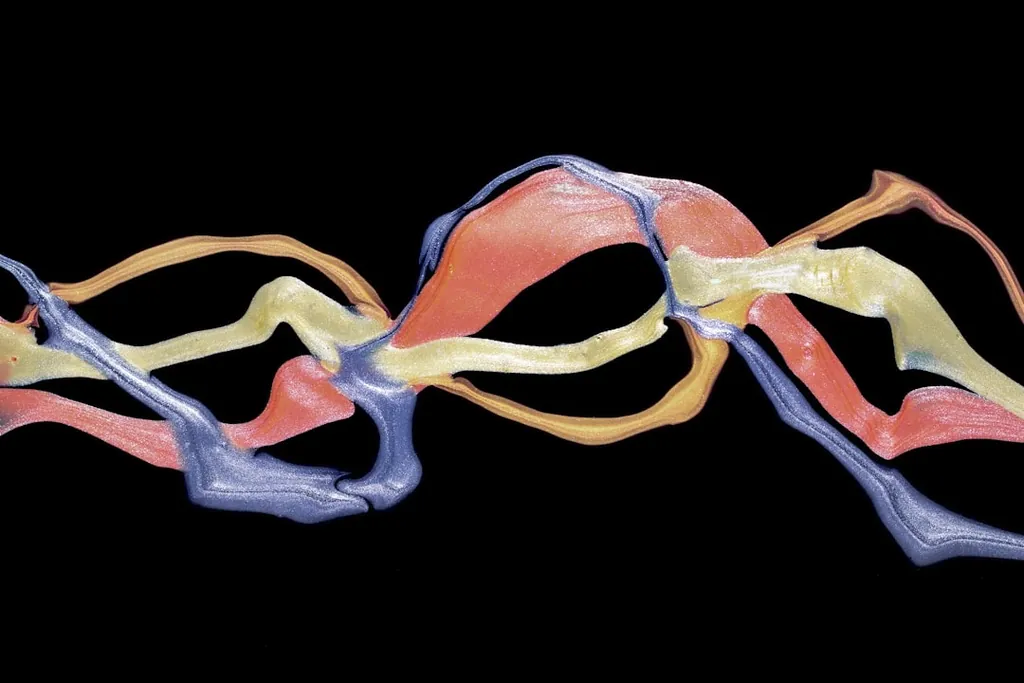
The defining structural finding for tirzepatide is that one peptide engages two distinct Class B GPCRs in similar overall conformation but with meaningfully different contact details. Those contact differences are what produce the biased pharmacology described in the next section. The 2022 cryo-EM paper resolves tirzepatide bound to GIPR-Gs and GLP-1R-Gs at 3.4 Å each.
Continuous α-helix at both receptors
In both bound structures the peptide adopts a single continuous α-helix. The N-terminus reaches deep into the seven-transmembrane bundle; the C-terminal half lies along the extracellular domain (ECD) of each receptor. Researchers familiar with the GLP-1 / GIP / glucagon agonist literature will recognize this overall pose. It's the same architecture seen in the retatrutide cryo-EM structures and in the dual GLP-1R/GCGR agonist literature.
N-terminal segment: residues 1–9
The N-terminal segment penetrates the receptor transmembrane core and engages the conserved Class B GPCR activation machinery at both targets. This region is largely conserved across the two receptors, which is why the same N-terminal sequence works in both pockets.
C-terminal half: the selectivity-tuning segment
From residue 10 onward, the peptide leaves the transmembrane core. It contacts the receptor extracellular domain, the extracellular tip of TM1, and ECL1. Side-chain orientations at ECL1 and ECL3 differ between GIPR-bound and GLP-1R-bound poses. These are the contacts the medicinal-chemistry team had to balance.
Activation conformations differ between the two receptors
All-atom molecular-dynamics simulations following the cryo-EM work track how each receptor's TM6 and ECL3 sample distinct conformations under tirzepatide binding. The MD work supports the cryo-EM-derived hypothesis that the same peptide produces different activation conformations at GIPR and GLP-1R — and that this difference is the structural origin of the biased signaling profile downstream.
Comparative potency and biased agonism
In cell-based receptor activation assays, tirzepatide doesn't behave identically at the two targets. The receptor-level pharmacology, characterized in the study by Willard and colleagues, is described as imbalanced and biased.
Imbalanced potency
At the human GIP receptor, tirzepatide engages with potency comparable to native GIP. At the human GLP-1 receptor, it is roughly 5-fold less potent than native GLP-1. Both observations are receptor-level and apply to in-vitro pharmacology only.
Biased signaling at GLP-1R
At GLP-1R, tirzepatide preferentially activates the cAMP / Gαs response over β-arrestin recruitment. Compared with native GLP-1, β-arrestin recruitment is markedly attenuated. The functional consequence in cell-based assays is reduced GLP-1R internalization and slower agonist-induced receptor desensitization — a more durable signaling profile across repeat-stimulation paradigms.
What the structural data predicts
The cryo-EM and MD work predicts exactly this kind of biased profile. The C-terminal contacts at ECL1 / ECL3 differ from the native GLP-1 binding pose, and the differential contact pattern is what tunes G-protein coupling efficiency relative to β-arrestin recruitment. Researchers reporting GLP-1R activation data for tirzepatide should pair Gαs / cAMP readouts with β-arrestin readouts to capture both halves of the bias.
Implications for assay design
Parallel-receptor assays should normalize each receptor's response against its own native ligand — not against a single common reference. Use GLP-1 alone as the reference and tirzepatide will look under-active at GIPR. Use GIP alone and it will look over-active at GLP-1R. Both interpretations are misleading. The cleanest reporting practice is per-receptor EC50 with the matched native-ligand control on the same plate, plus separate cAMP and β-arrestin readouts at GLP-1R to surface the bias.
Structural class context
Tirzepatide sits in an actively expanding structural class of multi-receptor peptide agonists built on the same general design language: GIP-derived backbone, Aib at the protease-cleavage site, lipidated C-terminal tail for albumin binding. Our retatrutide structural piece covers the triple-agonist extension of the same playbook — same backbone choice, same Aib2 strategy, same lipidation motif — with a third receptor (GCGR) added. Reading the two side by side highlights how the design space scales: each additional target receptor pushes the C-terminal segment to balance more contacts at once. Tirzepatide stops at two; retatrutide and NN1706 push to three.
Regulatory status
Tirzepatide is the first commercially approved dual-incretin receptor agonist. The FDA has cleared the compound under the brand names Mounjaro (with a type-2-diabetes indication) and Zepbound (with a chronic-weight-management indication). These are factual regulatory data points. Nothing here should be read as outcome claims about the indications themselves.
Research-grade vs. pharmaceutical-grade material
Optides supplies tirzepatide as a research-grade chemical for in-vitro work only. The chemistry — sequence, modifications, lipidation — matches the published structure characterized in the cryo-EM work, but research-grade material is not equivalent to and is not a substitute for the FDA-approved pharmaceutical product of the same chemical name. The distinction matters: the pharmaceutical product is manufactured under regulatory-grade quality controls and is intended for clinical use; research-grade material is intended exclusively for laboratory research. For broader context on the framework that shapes how research-grade peptides are sold and handled in the United States, see our research-use legal landscape article.
Frequently Asked Questions
What is tirzepatide and what does it target?
Tirzepatide (development code LY3298176, marketed as Mounjaro and Zepbound) is a synthetic 39-amino-acid peptide that engages two Class B GPCRs as a dual agonist: the GIP receptor (GIPR) and the GLP-1 receptor (GLP-1R). Cell-based pharmacology assays show concentration-responsive activity at both. Research-grade material sold for in-vitro work is not equivalent to and is not a substitute for the FDA-approved pharmaceutical product.
What modifications does tirzepatide carry and why?
Tirzepatide contains two α-aminoisobutyric acid (Aib) residues at positions 2 and 13. Aib2 introduces conformational rigidity at the N-terminal cleavage site of dipeptidyl peptidase 4 (DPP-4) and is the principal source of protease resistance. Aib13 supports activity at the GIP receptor. The peptide is acylated at lysine 20 with a γGlu-2×OEG linker bearing a C20 fatty diacid, which enables albumin binding and the long pharmacokinetic half-life seen in the literature.
What does "imbalanced and biased" mean for tirzepatide?
Imbalanced refers to potency: in cell-based receptor assays, tirzepatide engages GIPR with potency comparable to native GIP but is roughly 5-fold less potent than native GLP-1 at the GLP-1 receptor. Biased refers to signaling: at GLP-1R the molecule preferentially activates the cAMP / Gαs pathway over β-arrestin recruitment, which slows receptor internalization and agonist-induced desensitization. Both observations are receptor-level and apply to in-vitro pharmacology; the bundle does not extend them to therapeutic claims.
Has tirzepatide been approved for human use?
Yes — the FDA has approved tirzepatide as Mounjaro (for type-2 diabetes) and Zepbound (for chronic weight management). Optides supplies tirzepatide as a research-grade chemical for in-vitro work only; research-grade material is not equivalent to and is not a substitute for those approved pharmaceutical products. Researchers ordering material for laboratory studies should treat the chemistry separately from the regulatory product.
Conclusion
Tirzepatide is a structurally well-defined 39-residue peptide engineered from a GIP backbone, with α-aminoisobutyric acid at positions 2 (for DPP-4 resistance) and 13 (for GIPR activity), and a Lys20 fatty-diacid acylation that supports albumin binding. Cryo-EM evidence resolves the peptide bound to GIPR-Gs and GLP-1R-Gs as a continuous α-helix in both cases, with the N-terminal segment driving activation and the C-terminal contacts driving selectivity. The C-terminal differences are also what produce the imbalanced and biased pharmacology: comparable to native GIP at GIPR, lower-potency and Gαs-favoring at GLP-1R. The compound is FDA-approved as Mounjaro and Zepbound; research-grade material is a separate research-use category. For research use only.
For research use only. Not for human or animal consumption of any kind. The information in this article is for educational purposes only and is not intended to diagnose, treat, cure, or prevent any disease. The statements made have not been evaluated by the U.S. Food and Drug Administration. These products are NOT FDA APPROVED. Please consult with a licensed healthcare professional before making any decisions regarding your health or research.
Optides LLC is a chemical supplier. Optides LLC is not a compounding pharmacy or chemical compounding facility as defined under 503A of the Federal Food, Drug, and Cosmetic Act. Optides LLC is not an outsourcing facility as defined under 503B of the Federal Food, Drug, and Cosmetic Act.

